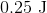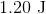Isothermic, Isobaric, and Adiabatic Processes
Help Questions
Physical Chemistry › Isothermic, Isobaric, and Adiabatic Processes
A sample of an ideal gas is initially at a volume of 



Explanation
The expression for the relationship between heat (


The work (


Plugging work (
Plugging the values given into the internal energy equation gives:
At 



Explanation
The process as described by the question is an isothermal process. An isothermal process has a constant temperature, therefore, there is no change in temperature. For an isothermal reversible process, the work done by the system is:
Plugging the values given into the equation gives:
Which of the following processes are considered an isothermic reaction?
I. Condensation
II. Evaporation
III. Sublimation
I, II and III
I only
II only
I and II
Explanation
Isothermic reactions are characterized as reactions that occur in constant temperature. Recall that phase changes (such as condensation, evaporation, etc.) occur at a constant temperature (melting point, boiling point,e etc.). Consider the following example. Water boils at 

Melting of a solid to a liquid is an example of what type of reaction?
More than one of these are true
Isothermic
Isovolemic
Isobaric
Explanation
Isothermic reactions occur in constant temperature, isovolemic reactions occur in constant volume and isobaric reactions occur in constant pressure. During phase changes, like melting, the temperature stays constant (at the melting point). The pressure will also likely remain constant because most reactions are carried out at atmospheric pressure (
The volume, on the other hand, changes. Recall that gases have the highest volume, solids have the smallest volume and liquids have intermediate volume. This means that melting converts a low volume solid to higher volume liquid (therefore, this is not an isovolemic reaction). As a side note, recall that water is the only exception to this rule. Ice cube (solid water) has higher volume than liquid water.
At 




Explanation
The process as described by the question is an isothermal process. An isothermal process has a constant temperature, therefore, there no change in temperature. For an isothermal reversible process, the work done by the system is:
Convert the grams to moles of argon:
Plugging the values given into the equation gives:
Which of the following is not true for an isothermal process?
Explanation
For an isothermal process there is no change in temperature, therefore, the temperature is a constant. An ideal gas has an internal energy, 
If you rearrange the equation, you will find that:
A simple explanation of this is that all the heat applied to the system is used to do the work. A piston is a good example for this phenomenon. This happens when every bit of energy applied or removed from a piston as work is done so slowly that all the heat has enough time to conduct into or out of the system and maintain a constant temperature.
A sample of an ideal gas is initially at a volume of 



Explanation
The expression for the relationship between heat (


The work (


Plugging work (
Plugging the values given into the internal energy equation gives: