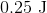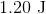Thermodynamics
Help Questions
Physical Chemistry › Thermodynamics
A sample of an ideal gas is initially at a volume of 



Explanation
The expression for the relationship between heat (


The work (


Plugging work (
Plugging the values given into the internal energy equation gives:
A sample of an ideal gas is initially at a volume of 



Explanation
The expression for the relationship between heat (


The work (


Plugging work (
Plugging the values given into the internal energy equation gives:
In an exergonic reaction, products will have __________ Gibbs free energy and the reaction is __________.
lower . . . spontaneous
lower . . . nonspontaneous
higher . . . spontaneous
higher . . . nonspontaneous
Explanation
Exergonic reaction suggests that the Gibbs free energy is negative. Since the change in Gibbs free energy is defined as Gibbs free energy of products - Gibbs free energy of reactants, a negative change in Gibbs free energy suggests that the products have a lower Gibbs free energy than reactants. A reaction is spontaneous if it has negative Gibbs free energy; therefore, exergonic reactions are always spontaneous. This is because the reaction is producing a more stable product (lower energy) from a less stable reactant (higher energy).
In an exergonic reaction, products will have __________ Gibbs free energy and the reaction is __________.
lower . . . spontaneous
lower . . . nonspontaneous
higher . . . spontaneous
higher . . . nonspontaneous
Explanation
Exergonic reaction suggests that the Gibbs free energy is negative. Since the change in Gibbs free energy is defined as Gibbs free energy of products - Gibbs free energy of reactants, a negative change in Gibbs free energy suggests that the products have a lower Gibbs free energy than reactants. A reaction is spontaneous if it has negative Gibbs free energy; therefore, exergonic reactions are always spontaneous. This is because the reaction is producing a more stable product (lower energy) from a less stable reactant (higher energy).
At 



Explanation
The process as described by the question is an isothermal process. An isothermal process has a constant temperature, therefore, there is no change in temperature. For an isothermal reversible process, the work done by the system is:
Plugging the values given into the equation gives:
At 



Explanation
The process as described by the question is an isothermal process. An isothermal process has a constant temperature, therefore, there is no change in temperature. For an isothermal reversible process, the work done by the system is:
Plugging the values given into the equation gives:
Which of the following processes are considered an isothermic reaction?
I. Condensation
II. Evaporation
III. Sublimation
I, II and III
I only
II only
I and II
Explanation
Isothermic reactions are characterized as reactions that occur in constant temperature. Recall that phase changes (such as condensation, evaporation, etc.) occur at a constant temperature (melting point, boiling point,e etc.). Consider the following example. Water boils at 

Which of the following processes are considered an isothermic reaction?
I. Condensation
II. Evaporation
III. Sublimation
I, II and III
I only
II only
I and II
Explanation
Isothermic reactions are characterized as reactions that occur in constant temperature. Recall that phase changes (such as condensation, evaporation, etc.) occur at a constant temperature (melting point, boiling point,e etc.). Consider the following example. Water boils at 

Which of the following is true regarding enthalpy and entropy?
The enthalpy of liquid water is nonzero whereas enthalpy of hydrogen gas is zero
The entropy of liquid water is higher than the entropy of hydrogen gas
Both of these are true
None of these are true
Explanation
Enthalpy is the amount of internal energy contained in a compound whereas entropy is the amount of intrinsic disorder within the compound. Enthalpy is zero for elemental compounds such hydrogen gas and oxygen gas; therefore, enthalpy is nonzero for water (regardless of phase). Entropy, or the amount of disorder, is always highest for gases and lowest for solids. This is because gas molecules are widely spread out and, therefore, are more disordered than solids and liquids. Hydrogen gas will have a higher entropy than liquid water.
According to the law of thermodynamics, which of the following statement(s) is/are true?
I. Enthalpy of a system is always increasing
II. Entropy of a system is always increasing
III. Absolute entropy can never be negative
III only
II only
I and II
II and III
Explanation
The first law of thermodynamics states that the energy of the universe is always constant, which implies that energy cannot be created or destroyed. The energy lost by a system is gained by surroundings and vice versa; however, the total energy of the universe is always constant. The second law of thermodynamics states that the entropy, or the amount of disorder in the universe, is always increasing. This suggests that the universe is always going towards a more disordered state. Based on these two laws, we can determine that statement I and statement II are false. Note that these two statements are talking about the system, rather than the universe. The energy (in the form of enthalpy) and entropy can increase or decrease in a system. The surroundings will compensate accordingly to keep the energy of universe constant and increase the entropy. Absolute entropy of a system, surroundings or the universe can never be negative because it isn’t possible to have negative disorder (this is due to the definition of entropy; just remember that entropy can never be negative). Note that the change in entropy can, however, be negative.