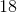The Periodic Table
Help Questions
GED Science › The Periodic Table
On the periodic table, carbon is atomic number 

Explanation
The standard sub-atomic particles are protons, neutrons, and electrons. Protons and neutrons are found in the nucleus, while electrons move freely around the nucleus. This question is specifically asking about the number of protons and neutrons in an atom of carbon.
Each proton and each neutron has a mass of approximately 

The atomic number refers to the number of protons in the nucleus of the atom. Since the atomic number of carbon is six, there are six protons in the nucleus. For this question, however, we need the total of both the protons and neutrons (not just the protons).