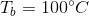All Physical Chemistry Resources
Example Questions
Example Question #1 : Colligative Properties
What is the boiling point of a 100mL solution of water after the addition of 29g of sodium chloride?
Possible Answers:
Correct answer:
Explanation:
We can use the boiling point elevation equation in order to find the new boiling point for the solution.
The change in temperature will be equal to the boiling point elevation constant multiplied by the molality of the solution multiplied by the van't Hoff factor. The van't Hoff factor is a variable that incorporates how many ions the added solute will dissolve into when in solution. Sodium chloride will dissolve into two ions, so the van't Hoff factor is 2.
This means that the boiling point of the water has been elevated by 5.2 degrees, meaning that the boiling point of the solution is 105.2 degrees celsius.
Courtney
Certified Tutor
Certified Tutor
Ohio University-Main Campus, Bachelor of Science, Science Teacher Education.
Paige
Certified Tutor
Certified Tutor
Colorado School of Mines, Bachelor of Science, Chemical and Biomolecular Engineering.
All Physical Chemistry Resources
Physical Chemistry Tutors in Top Cities:
Atlanta Physical Chemistry Tutors, Austin Physical Chemistry Tutors, Boston Physical Chemistry Tutors, Chicago Physical Chemistry Tutors, Dallas Fort Worth Physical Chemistry Tutors, Denver Physical Chemistry Tutors, Houston Physical Chemistry Tutors, Kansas City Physical Chemistry Tutors, Los Angeles Physical Chemistry Tutors, Miami Physical Chemistry Tutors, New York City Physical Chemistry Tutors, Philadelphia Physical Chemistry Tutors, Phoenix Physical Chemistry Tutors, San Diego Physical Chemistry Tutors, San Francisco-Bay Area Physical Chemistry Tutors, Seattle Physical Chemistry Tutors, St. Louis Physical Chemistry Tutors, Tucson Physical Chemistry Tutors, Washington DC Physical Chemistry Tutors
Popular Courses & Classes
GRE Courses & Classes in Denver, SAT Courses & Classes in Boston, SSAT Courses & Classes in Atlanta, ACT Courses & Classes in Denver, GRE Courses & Classes in Chicago, SSAT Courses & Classes in Chicago, GMAT Courses & Classes in Philadelphia, ACT Courses & Classes in Los Angeles, MCAT Courses & Classes in Washington DC, ISEE Courses & Classes in Los Angeles
Popular Test Prep
GRE Test Prep in Los Angeles, ISEE Test Prep in Denver, GMAT Test Prep in Seattle, GMAT Test Prep in Philadelphia, SAT Test Prep in Philadelphia, GRE Test Prep in Dallas Fort Worth, MCAT Test Prep in Boston, GMAT Test Prep in New York City, MCAT Test Prep in Washington DC, SAT Test Prep in Miami