Carbonyl Products
Help Questions
Organic Chemistry › Carbonyl Products

What is the product of the given reaction?

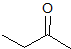



Explanation
The hydrolyzation of a nitrile with hydronium leads to a carboxylic acid. Only one choice is a carboxylic acid.

What is the product of the given reaction?





Explanation
The hydrolyzation of a nitrile with hydronium leads to a carboxylic acid. Only one choice is a carboxylic acid.

What is the product of the given reaction?





Explanation
This question tests knowledge on carboxylic acid derivatives and the formation of the carboxylic acid derivatives. 




What is the product of the given reaction?





Explanation
This question tests knowledge on carboxylic acid derivatives and the formation of the carboxylic acid derivatives. 



What is the product of a hydroboration–oxidation reaction with 1-hexylcyclohexene?
2-hexylcyclohexanol
1-hexylcyclohexanol
3-hexylcyclohexanol
Hexylcyclohexane
Cyclohexane
Explanation
This reaction is an electrophilic addition reaction with an alkene. This is one of many alkene addition reactions that can add an -OH group onto your starting material. The key aspect of an hydroboration-oxidation reaction is the anti-Markovinikov addition to the double bond. The -OH group should be on the least substituted of the two carbons that originate from the double bond. In light of this information, the answer is 2-cyclohexanol.
What is the product of a hydroboration–oxidation reaction with 1-hexylcyclohexene?
2-hexylcyclohexanol
1-hexylcyclohexanol
3-hexylcyclohexanol
Hexylcyclohexane
Cyclohexane
Explanation
This reaction is an electrophilic addition reaction with an alkene. This is one of many alkene addition reactions that can add an -OH group onto your starting material. The key aspect of an hydroboration-oxidation reaction is the anti-Markovinikov addition to the double bond. The -OH group should be on the least substituted of the two carbons that originate from the double bond. In light of this information, the answer is 2-cyclohexanol.
Which combination of starting material and reagent will produce the product shown?

5-hydroxyvaleric acid and
Pentanedioic acid and
4-hydroxybutyric acid and
4-hydroxybutyric acid and
5-hydroxyvaleric acid and
Explanation
5-hydroxyvaleric acid (5-hydroxypentanoic acid) is a five-carbon carboxylic acid alcohol and will produce the required lactone. This reaction is typically acid-catalyzed. Pentanedioic acid (glutaric acid) is a linear dicarboxylic acid, 4-hydroxybutyric acid only has four carbons and will not produce the desired product without being modified.
Which combination of starting material and reagent will produce the product shown?

5-hydroxyvaleric acid and
Pentanedioic acid and
4-hydroxybutyric acid and
4-hydroxybutyric acid and
5-hydroxyvaleric acid and
Explanation
5-hydroxyvaleric acid (5-hydroxypentanoic acid) is a five-carbon carboxylic acid alcohol and will produce the required lactone. This reaction is typically acid-catalyzed. Pentanedioic acid (glutaric acid) is a linear dicarboxylic acid, 4-hydroxybutyric acid only has four carbons and will not produce the desired product without being modified.
An organic chemist wants to synthesize an ether product. She begins with methanol and ethanol as her substrates. How would she go about synthesizing the desired ether product?
Convert the alcohol on the methanol to a tosylate group and allow this modified substrate to react with the ethanol
It is impossible to create an ether product with these substrates
Put both substrates in an aprotic solvent and allow them to react via
Add heat to the substrates to elicit a dehydration reaction
Explanation
The substrates must be modified in order to attain the desired product. In order to get the desired product, she needs to change one of the alcohol groups into a good leaving group. One way to do this is to react the methanol with 
An organic chemist wants to synthesize an ether product. She begins with methanol and ethanol as her substrates. How would she go about synthesizing the desired ether product?
Convert the alcohol on the methanol to a tosylate group and allow this modified substrate to react with the ethanol
It is impossible to create an ether product with these substrates
Put both substrates in an aprotic solvent and allow them to react via
Add heat to the substrates to elicit a dehydration reaction
Explanation
The substrates must be modified in order to attain the desired product. In order to get the desired product, she needs to change one of the alcohol groups into a good leaving group. One way to do this is to react the methanol with 





