Support Reaction Claims With Evidence
Help Questions
Chemistry › Support Reaction Claims With Evidence
A student claims: "Iron rusted because it reacted with oxygen in the air."
Evidence:
-
After a week, a reddish-brown coating formed on the iron nail.
-
The nail was left outdoors where it rained twice.
-
The nail was attracted to a magnet both before and after the week.
-
The mass of the nail increased from 5.00 g to 5.08 g (after drying).
Which evidence is most directly relevant to the claim that iron reacted with oxygen?
Evidence 2 and 3, because outdoor conditions and magnetism are enough to prove a reaction with oxygen.
Evidence 1 and 4, because a rust-colored coating and a mass increase are consistent with oxygen combining with iron.
Evidence 3, because magnetism proves oxygen was present.
Evidence 2, because rain is the main reactant in rusting.
Explanation
This question tests your ability to evaluate whether evidence adequately supports scientific claims about chemical reactions, distinguishing relevant from irrelevant evidence and assessing whether evidence is sufficient for the claim. Supporting scientific claims requires three things: (1) RELEVANCE—does the evidence actually relate to the claim? If claiming "iron reacted with oxygen," evidence about mass increase and rust formation is relevant, but evidence about magnetism is not directly related to oxygen reaction. (2) SUFFICIENCY—is there enough evidence? Multiple indicators that specifically point to oxygen addition provide sufficient support. (3) QUALITY—is the evidence specific and measurable? "Mass increased from 5.00 g to 5.08 g" is stronger than "got heavier." Evidence 1 (rust-colored coating) and Evidence 4 (mass increase) both directly support oxygen reaction—rust (iron oxide) contains oxygen atoms that weren't in pure iron, and the mass increase (0.08 g) represents oxygen atoms that combined with iron atoms to form the rust compound. Choice C correctly evaluates the evidence by identifying that the characteristic rust color indicates iron oxide formation and the mass increase quantitatively shows matter (oxygen) was added to the iron. Choice A incorrectly suggests rain is the main reactant (water accelerates rusting but oxygen is the reactant), while Choice B misinterprets magnetism as proving oxygen presence. The evidence evaluation framework: (1) Read the claim carefully: iron reacted with oxygen. (2) For EACH piece of evidence, ask: Does rust color indicate iron oxide? YES—rust is iron-oxygen compound. Does mass increase indicate oxygen addition? YES—extra mass must come from somewhere (oxygen from air). Does rain prove oxygen reaction? NO—water helps but isn't the reactant. Does magnetism prove oxygen reaction? NO—many iron compounds are magnetic. (3) Count relevant evidence: Two direct indicators (rust appearance + mass gain) = strong support. (4) Make judgment: Physical and quantitative evidence of oxygen addition = claim well supported. Evidence quality checklist: STRONG evidence includes specific color (reddish-brown = iron oxide), quantitative data (0.08 g increase), and direct relevance to oxygen combination (mass increase = oxygen atoms added).
A student claims: "Mixing solutions of silver nitrate and sodium chloride causes a chemical reaction (a precipitate forms)."
Evidence/observations:
-
A cloudy white solid appears immediately after mixing.
-
The beaker feels slightly cooler to the touch.
-
When the mixture is filtered, a white solid remains on the filter paper.
-
The original silver nitrate solution was clear and colorless.
-
The total volume after mixing is about 100 mL.
Which pieces of evidence are most directly relevant to supporting the claim that a precipitate formed due to a chemical reaction?
1 and 3 only
2 and 4 only
4 and 5 only
1, 2, 3, 4, and 5 (all of them)
Explanation
This question tests your ability to evaluate whether evidence adequately supports scientific claims about chemical reactions, distinguishing relevant from irrelevant evidence and assessing whether evidence is sufficient for the claim. Supporting scientific claims requires three things: (1) RELEVANCE—does the evidence actually relate to the claim? If claiming "reaction is exothermic," evidence about temperature increase is relevant, but evidence about color is not (color doesn't indicate exo vs endo). (2) SUFFICIENCY—is there enough evidence? Single weak observation usually insufficient; multiple strong observations or reproducible quantitative data provide sufficient support. (3) QUALITY—is the evidence specific and measurable? "Temperature increased by 12°C" is stronger than "it got warm." For claim "chemical reaction occurred," relevant sufficient evidence might include gas production + temperature increase + precipitate formation (three indicators of new substances), while just "substances mixed" would be insufficient (mixing doesn't prove reaction). For this claim of a precipitate-forming reaction, evidence 1 (cloudy white solid) and 3 (white solid on filter) are directly relevant as they show insoluble product formation, while 2 (cooler) might indicate endothermic but not precipitate, 4 (original clear) is baseline, and 5 (volume) is irrelevant. Choice A correctly evaluates the evidence by selecting only 1 and 3 as most directly relevant to precipitate formation, ensuring focus on sufficiency for that specific claim. Options like C include irrelevant pieces, which dilutes support—remember, quality means prioritizing direct evidence! The evidence evaluation framework: (1) Read the claim carefully: What exactly is being claimed? (2) For EACH piece of evidence, ask: Does this relate to the claim? (Relevance test), Does this indicate what the claim says? (Support test), Could this observation happen WITHOUT the claim being true? (Alternative explanation test). (3) Count relevant evidence: How many pieces directly support the claim? Is it one weak observation or multiple strong indicators? (4) Make judgment: If multiple relevant, strong pieces of evidence with no contradictions = claim well supported. If only weak or ambiguous evidence = claim not well supported. If evidence contradicts claim = claim not supported. Be honest about evidence quality! Evidence quality checklist: STRONG evidence is (1) Specific ("bubbles formed" not "something happened"), (2) Quantitative when possible ("temperature rose 15°C" better than "got hot"), (3) Reproducible ("happened in all three trials" better than "happened once"), (4) Directly relevant (addresses the claim directly), (5) Not explainable by alternatives (gas from reaction, not from boiling). WEAK evidence is vague, qualitative only, single occurrence, tangentially related, or explainable other ways. For claim "Substance X reacts with acid," strong evidence: "X dissolved in acid with vigorous bubbling and 20°C temperature rise in repeated trials." Weak evidence: "X and acid were mixed." Use strong evidence to support claims!
A student claims: "A reaction produced a gas when liquid A was added to solid B."
Evidence:
-
The balloon placed over the flask inflated during the first minute.
-
The mass of the closed flask (with balloon) stayed the same: 152.34 g before and 152.34 g after.
-
The mixture changed from colorless to pale yellow.
-
The student used a larger balloon in Trial 2 than in Trial 1.
Which evidence is most direct for supporting the claim that a gas was produced?
Evidence 4, because changing balloon size causes gas to be produced.
Evidence 1, because balloon inflation indicates gas formation.
Evidence 3, because any color change means a gas formed.
Evidence 2, because constant mass proves a gas was produced.
Explanation
This question tests your ability to evaluate whether evidence adequately supports scientific claims about chemical reactions, distinguishing relevant from irrelevant evidence and assessing whether evidence is sufficient for the claim. Supporting scientific claims requires three things: (1) RELEVANCE—does the evidence actually relate to the claim? If claiming "reaction produced a gas," evidence about gas collection or pressure increase is relevant, but evidence about balloon size choice is not. (2) SUFFICIENCY—is there enough evidence? Direct observation of gas production provides sufficient support. (3) QUALITY—is the evidence specific and measurable? "Balloon inflated during first minute" is stronger than "something happened." Evidence 1 directly shows gas production—the balloon inflated because gas produced in the flask created pressure that pushed into the balloon, providing clear, visual proof of gas formation, while Evidence 2 supports but doesn't prove gas (mass conservation occurs whether gas forms or not), and Evidence 3 and 4 are irrelevant to gas production. Choice A correctly evaluates the evidence by identifying balloon inflation as the most direct indicator—gases occupy space and create pressure, so balloon inflation specifically indicates gas was produced inside the flask. Choice B misinterprets mass conservation (which occurs in all reactions), while Choices C and D confuse unrelated observations with gas production. The evidence evaluation framework: (1) Read the claim carefully: reaction produced a gas. (2) For EACH piece of evidence, ask: Does balloon inflation indicate gas? YES—gas fills balloon directly. Does constant mass prove gas? NO—mass conserved regardless. Does color change indicate gas? NO—color and gas formation are independent. Does balloon size matter? NO—any inflation shows gas. (3) Identify most direct evidence: Balloon inflation = gas pushing outward. (4) Make judgment: Direct physical evidence of gas presence = strong support. Evidence quality checklist: DIRECT evidence shows the phenomenon itself (balloon inflates = gas present), while INDIRECT evidence might be consistent with the claim but doesn't prove it (mass conservation happens with or without gas). Always prefer direct over indirect evidence!
A student claims: "A precipitate formed because a chemical reaction occurred when two clear solutions were mixed."
Evidence:
-
The mixture turned cloudy and a white solid appeared within 10 seconds.
-
After filtering, the solid remained on the filter paper and did not dissolve when rinsed with water.
-
The total volume of liquid in the beaker increased by 2 mL because the student added extra water.
-
The room lights were turned off during one trial.
Which piece of evidence does NOT support the claim?
Evidence 4
Evidence 2
Evidence 3
Evidence 1
Explanation
This question tests your ability to evaluate whether evidence adequately supports scientific claims about chemical reactions, distinguishing relevant from irrelevant evidence and assessing whether evidence is sufficient for the claim. Supporting scientific claims requires three things: (1) RELEVANCE—does the evidence actually relate to the claim? If claiming "precipitate formed because of chemical reaction," evidence about solid formation and properties is relevant, but evidence about room lighting is not. (2) SUFFICIENCY—is there enough evidence? Multiple observations about the precipitate provide sufficient support. (3) QUALITY—is the evidence specific and measurable? "White solid appeared within 10 seconds" is stronger than "something formed." Evidence 1, 2, and 3 all relate to the precipitate or reaction process: cloudiness and white solid formation indicate new substance creation, insolubility confirms it's a true precipitate (not just undissolved reactant), and volume change relates to the mixing process—but Evidence 4 about room lights has no relevance to whether a chemical reaction occurred or precipitate formed. Choice D correctly identifies that room lighting is completely irrelevant to the chemical process—reactions occur the same way in light or dark, and precipitate formation doesn't depend on external lighting conditions. All other choices incorrectly select evidence that actually supports or relates to the claim. The evidence evaluation framework: (1) Read the claim carefully: precipitate formed BECAUSE of chemical reaction. (2) For EACH piece of evidence, ask: Does white solid formation support the claim? YES—new solid indicates reaction. Does insolubility support precipitate claim? YES—true precipitates don't redissolve easily. Does volume change matter? SOMEWHAT—relates to experimental procedure. Do room lights affect reactions? NO—completely irrelevant. (3) Identify irrelevant evidence: Lighting conditions don't affect chemical processes. (4) Make judgment: Evidence 4 provides no support whatsoever. Evidence quality checklist: RELEVANT evidence must connect to the claim (solid formation connects to precipitate claim), while IRRELEVANT evidence has no logical connection (room lighting doesn't affect molecular interactions). When evaluating evidence, always ask: "How does this relate to what's being claimed?"
A company claims: "Our new cleaner chemically removes rust from iron."
Evidence provided:
-
Before/after photos show a rusty nail looks shiny after soaking for 10 minutes.
-
A customer testimonial says, "It worked instantly!"
-
In a class test, the mass of a rusty nail decreases from 5.20 g to 5.05 g after soaking and rinsing.
-
The cleaner has a lemon scent.
Which evidence is strongest and most relevant for supporting the claim that a chemical change (rust removal) occurred?
(2), because personal experience is the most reliable scientific evidence.
(4), because scent shows the cleaner contains reactive chemicals.
(3), because a measured mass change after treatment is quantitative evidence consistent with rust being removed from the nail.
(1), because photos alone prove a chemical reaction happened.
Explanation
This question tests your ability to evaluate whether evidence adequately supports scientific claims about chemical reactions, distinguishing relevant from irrelevant evidence and assessing whether evidence is sufficient for the claim. Supporting scientific claims requires three things: (1) RELEVANCE—does the evidence actually relate to the claim? If claiming "chemically removes rust," evidence about mass change or rust disappearance is relevant, but evidence about scent is not. (2) SUFFICIENCY—is there enough evidence? Single weak observation usually insufficient; multiple strong observations or reproducible quantitative data provide sufficient support. (3) QUALITY—is the evidence specific and measurable? "Mass decreased by 0.15 g" is stronger than "it looked cleaner." For the claim about chemical rust removal, observation 3 provides the strongest evidence: the measured mass decrease (5.20 g → 5.05 g) is quantitative proof that material (rust) was removed from the nail. Photos (1) show visual change but could be surface cleaning, testimonials (2) are subjective and vague, and lemon scent (4) is irrelevant to rust removal mechanism. Choice D correctly identifies that measured mass change after treatment is quantitative evidence consistent with rust being removed—this objective measurement proves material was actually removed, not just visually altered. Choice A irrelevantly focuses on scent; Choice B wrongly elevates anecdotal evidence; Choice C overstates what photos alone can prove without supporting data. The evidence evaluation framework: (1) Read the claim carefully: Does the cleaner chemically remove rust? (2) For EACH piece of evidence, ask: Do photos prove chemical change? No—could be physical cleaning. Is testimonial scientific evidence? No—subjective and vague. Does mass decrease indicate rust removal? Yes—quantitative proof material removed. Does scent indicate rust removal? No—just product characteristic. (3) Count relevant evidence: One strong quantitative measurement supporting removal. (4) Make judgment: Mass change provides objective support for claim. Evidence quality checklist: The mass change evidence is STRONG because it's (1) Quantitative (0.15 g decrease), (2) Measurable (using balance), (3) Objective (not opinion-based), (4) Directly relevant (mass loss = material removed), (5) Consistent with claim (rust removal would decrease mass). Photos and testimonials are WEAK—subjective and not quantitative. Use measurable changes as primary evidence!
A student claims: "The reaction between magnesium metal and hydrochloric acid produces hydrogen gas."
In a lab, the student places a 2.0 cm strip of magnesium into 50 mL of 1.0 M HCl at room temperature and records these observations:
-
Bubbles form rapidly on the magnesium surface.
-
A burning splint held near the mouth of the test tube makes a brief "pop" sound.
-
The solution temperature increases from 22.0°C to 27.5°C.
-
The magnesium strip becomes smaller over 2 minutes.
Which statement best evaluates whether the evidence supports the claim?
The evidence supports the claim mainly because the solution warmed up, which proves hydrogen formed.
The evidence supports the claim because the pop test is a specific indicator of hydrogen gas, and bubbling shows gas production.
The evidence does not support the claim because a temperature increase does not relate to gas identity.
The evidence is insufficient because only the magnesium shrinking is relevant to identifying the gas.
Explanation
This question tests your ability to evaluate whether evidence adequately supports scientific claims about chemical reactions, distinguishing relevant from irrelevant evidence and assessing whether evidence is sufficient for the claim. Supporting scientific claims requires three things: (1) RELEVANCE—does the evidence actually relate to the claim? If claiming "reaction is exothermic," evidence about temperature increase is relevant, but evidence about color is not (color doesn't indicate exo vs endo). (2) SUFFICIENCY—is there enough evidence? Single weak observation usually insufficient; multiple strong observations or reproducible quantitative data provide sufficient support. (3) QUALITY—is the evidence specific and measurable? "Temperature increased by 12°C" is stronger than "it got warm." For claim "chemical reaction occurred," relevant sufficient evidence might include gas production + temperature increase + precipitate formation (three indicators of new substances), while just "substances mixed" would be insufficient (mixing doesn't prove reaction). In this case, the claim is that magnesium and HCl produce hydrogen gas, so relevant evidence includes bubbling (gas production), the pop test (specific for hydrogen), temperature increase (exothermic reaction indicator), and magnesium shrinking (metal being consumed), while all observations are related but the pop test and bubbling are most direct for gas identity. Choice B correctly evaluates the evidence by identifying the pop test as a specific indicator for hydrogen and bubbling as proof of gas production, collectively providing strong, sufficient support for the claim. Choices like A fail by incorrectly dismissing relevant evidence like temperature, which supports a reaction occurring, while D overemphasizes temperature as proving hydrogen specifically, which it doesn't—keep focusing on what each piece truly indicates! The evidence evaluation framework: (1) Read the claim carefully: What exactly is being claimed? (2) For EACH piece of evidence, ask: Does this relate to the claim? (Relevance test), Does this indicate what the claim says? (Support test), Could this observation happen WITHOUT the claim being true? (Alternative explanation test). (3) Count relevant evidence: How many pieces directly support the claim? Is it one weak observation or multiple strong indicators? (4) Make judgment: If multiple relevant, strong pieces of evidence with no contradictions = claim well supported. If only weak or ambiguous evidence = claim not well supported. If evidence contradicts claim = claim not supported. Be honest about evidence quality! Evidence quality checklist: STRONG evidence is (1) Specific ("bubbles formed" not "something happened"), (2) Quantitative when possible ("temperature rose 15°C" better than "got hot"), (3) Reproducible ("happened in all three trials" better than "happened once"), (4) Directly relevant (addresses the claim directly), (5) Not explainable by alternatives (gas from reaction, not from boiling). WEAK evidence is vague, qualitative only, single occurrence, tangentially related, or explainable other ways. For claim "Substance X reacts with acid," strong evidence: "X dissolved in acid with vigorous bubbling and 20°C temperature rise in repeated trials." Weak evidence: "X and acid were mixed." Use strong evidence to support claims!
A student claims: "Increasing temperature increases the rate of the reaction between an effervescent tablet and water."
The student drops identical tablets into 100 mL of water and measures the time until fizzing stops.
Do the data support the claim?
Data:
- 10°C water: fizzing stops after 180 s
- 25°C water: fizzing stops after 95 s
- 40°C water: fizzing stops after 55 s
- 60°C water: fizzing stops after 50 s
Which statement best evaluates the claim using the data?
No, the data do not support the claim because the reaction time is not exactly cut in half each time temperature increases.
Yes, the data support the claim only because the tablet makes bubbles at all temperatures.
Yes, overall the data support the claim because higher temperature generally corresponds to shorter reaction time (faster rate), though the change from 40°C to 60°C is small.
No, the data contradict the claim because the reaction at 60°C is slower than at 40°C.
Explanation
This question tests your ability to evaluate whether evidence adequately supports scientific claims about chemical reactions, distinguishing relevant from irrelevant evidence and assessing whether evidence is sufficient for the claim. Supporting scientific claims requires three things: (1) RELEVANCE—does the evidence actually relate to the claim? If claiming "reaction is exothermic," evidence about temperature increase is relevant, but evidence about color is not (color doesn't indicate exo vs endo). (2) SUFFICIENCY—is there enough evidence? Single weak observation usually insufficient; multiple strong observations or reproducible quantitative data provide sufficient support. (3) QUALITY—is the evidence specific and measurable? "Temperature increased by 12°C" is stronger than "it got warm." For claim "chemical reaction occurred," relevant sufficient evidence might include gas production + temperature increase + precipitate formation (three indicators of new substances), while just "substances mixed" would be insufficient (mixing doesn't prove reaction). Here, the claim is that increasing temperature boosts reaction rate, and the data show reaction times decreasing from 180 s at 10°C to 50 s at 60°C, with quantitative measurements across multiple temperatures supporting faster rates at higher temps, though the fizzing itself is a general indicator. Choice A correctly evaluates the evidence by noting the trend of shorter times with higher temperatures, providing sufficient quantitative support despite the small change from 40°C to 60°C. Distractors like B fail by demanding an exact halving, which isn't required for support—science values trends, not perfection, so look for overall patterns! The evidence evaluation framework: (1) Read the claim carefully: What exactly is being claimed? (2) For EACH piece of evidence, ask: Does this relate to the claim? (Relevance test), Does this indicate what the claim says? (Support test), Could this observation happen WITHOUT the claim being true? (Alternative explanation test). (3) Count relevant evidence: How many pieces directly support the claim? Is it one weak observation or multiple strong indicators? (4) Make judgment: If multiple relevant, strong pieces of evidence with no contradictions = claim well supported. If only weak or ambiguous evidence = claim not well supported. If evidence contradicts claim = claim not supported. Be honest about evidence quality! Evidence quality checklist: STRONG evidence is (1) Specific ("bubbles formed" not "something happened"), (2) Quantitative when possible ("temperature rose 15°C" better than "got hot"), (3) Reproducible ("happened in all three trials" better than "happened once"), (4) Directly relevant (addresses the claim directly), (5) Not explainable by alternatives (gas from reaction, not from boiling). WEAK evidence is vague, qualitative only, single occurrence, tangentially related, or explainable other ways. For claim "Substance X reacts with acid," strong evidence: "X dissolved in acid with vigorous bubbling and 20°C temperature rise in repeated trials." Weak evidence: "X and acid were mixed." Use strong evidence to support claims!
A student claims: "Copper metal reacts with dilute hydrochloric acid to produce hydrogen gas."
The student places a clean copper strip into 50 mL of 1.0 M HCl at 25°C and observes:
-
No bubbles are seen after 10 minutes.
-
The copper strip remains shiny and appears unchanged.
-
The temperature stays at 25.0°C.
-
The hydrochloric acid has a strong smell.
Is the claim supported by the evidence? Choose the best evaluation.
No; the observations provide no evidence of gas production or reaction under these conditions, so the claim is not supported.
Yes; if the copper is shiny, it must be reacting quickly.
Yes; the strong smell of hydrochloric acid is evidence that hydrogen gas is being produced.
No; the claim is supported only if the solution changes color, and no color change was reported.
Explanation
This question tests your ability to evaluate whether evidence adequately supports scientific claims about chemical reactions, distinguishing relevant from irrelevant evidence and assessing whether evidence is sufficient for the claim. Supporting scientific claims requires three things: (1) RELEVANCE—does the evidence actually relate to the claim? If claiming "reaction is exothermic," evidence about temperature increase is relevant, but evidence about color is not (color doesn't indicate exo vs endo). (2) SUFFICIENCY—is there enough evidence? Single weak observation usually insufficient; multiple strong observations or reproducible quantitative data provide sufficient support. (3) QUALITY—is the evidence specific and measurable? "Temperature increased by 12°C" is stronger than "it got warm." For claim "chemical reaction occurred," relevant sufficient evidence might include gas production + temperature increase + precipitate formation (three indicators of new substances), while just "substances mixed" would be insufficient (mixing doesn't prove reaction). The claim states copper produces hydrogen with HCl, but evidence shows no bubbles, no change in copper or temperature, only HCl smell (which is baseline), lacking any reaction indicators. Choice C correctly evaluates the evidence by pointing out the absence of gas or reaction signs, meaning the claim lacks support. Distractors like A misattribute smell to hydrogen—stay encouraged, as recognizing no evidence is key to science! The evidence evaluation framework: (1) Read the claim carefully: What exactly is being claimed? (2) For EACH piece of evidence, ask: Does this relate to the claim? (Relevance test), Does this indicate what the claim says? (Support test), Could this observation happen WITHOUT the claim being true? (Alternative explanation test). (3) Count relevant evidence: How many pieces directly support the claim? Is it one weak observation or multiple strong indicators? (4) Make judgment: If multiple relevant, strong pieces of evidence with no contradictions = claim well supported. If only weak or ambiguous evidence = claim not well supported. If evidence contradicts claim = claim not supported. Be honest about evidence quality! Evidence quality checklist: STRONG evidence is (1) Specific ("bubbles formed" not "something happened"), (2) Quantitative when possible ("temperature rose 15°C" better than "got hot"), (3) Reproducible ("happened in all three trials" better than "happened once"), (4) Directly relevant (addresses the claim directly), (5) Not explainable by alternatives (gas from reaction, not from boiling). WEAK evidence is vague, qualitative only, single occurrence, tangentially related, or explainable other ways. For claim "Substance X reacts with acid," strong evidence: "X dissolved in acid with vigorous bubbling and 20°C temperature rise in repeated trials." Weak evidence: "X and acid were mixed." Use strong evidence to support claims!
A student claims: "Increasing acid concentration increases the reaction rate between magnesium ribbon and hydrochloric acid."
In each trial, the same length of magnesium ribbon was placed into $50.0,\text{mL}$ of HCl at $25.0^\circ\text{C}$. The time for the magnesium to completely disappear was recorded.
Do the data support the claim?
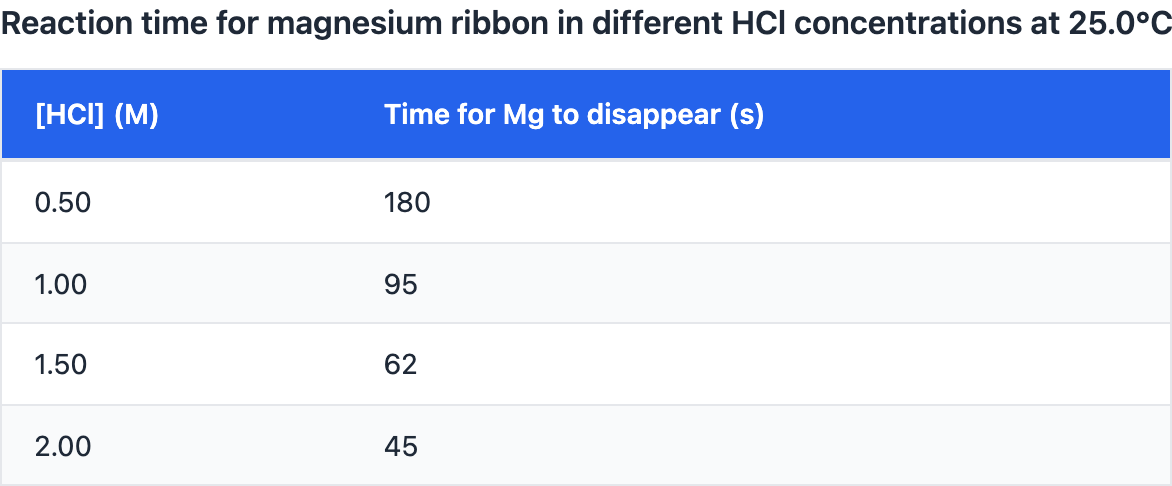
Yes. The claim is supported because magnesium is a reactive metal, so it will always react quickly regardless of concentration.
No. The data do not support the claim because the volume of acid is $50.0,\text{mL}$ in every trial.
No. The data show no relationship because the times are all different and not identical.
Yes. As HCl concentration increases, the time decreases, indicating a faster reaction rate.
Explanation
This question tests your ability to evaluate whether evidence adequately supports scientific claims about chemical reactions, distinguishing relevant from irrelevant evidence and assessing whether evidence is sufficient for the claim. Supporting scientific claims requires three things: (1) RELEVANCE—does the evidence actually relate to the claim? If claiming "reaction is exothermic," evidence about temperature increase is relevant, but evidence about color is not (color doesn't indicate exo vs endo). (2) SUFFICIENCY—is there enough evidence? Single weak observation usually insufficient; multiple strong observations or reproducible quantitative data provide sufficient support. (3) QUALITY—is the evidence specific and measurable? "Temperature increased by 12°C" is stronger than "it got warm." For claim "chemical reaction occurred," relevant sufficient evidence might include gas production + temperature increase + precipitate formation (three indicators of new substances), while just "substances mixed" would be insufficient (mixing doesn't prove reaction). The data table clearly shows that as HCl concentration increases from 0.5 M to 2.0 M, the time for magnesium to disappear decreases from 180 s to 45 s—this inverse relationship between concentration and reaction time directly supports the claim that increasing acid concentration increases reaction rate (faster reaction = less time needed). Choice A correctly evaluates this evidence by recognizing that decreasing time indicates faster reaction rate, establishing the clear relationship between concentration and rate. Choice B incorrectly focuses on the constant volume (50.0 mL) as if this invalidates the claim, when actually keeping volume constant while varying concentration is proper experimental design; Choice C misunderstands that different times are exactly what we expect when testing different concentrations; Choice D makes an irrelevant general statement about magnesium reactivity that doesn't address the specific concentration-rate relationship being tested. The evidence evaluation framework: (1) Read the claim carefully: What exactly is being claimed? (2) For EACH piece of evidence, ask: Does this relate to the claim? (Relevance test), Does this indicate what the claim says? (Support test), Could this observation happen WITHOUT the claim being true? (Alternative explanation test). (3) Count relevant evidence: How many pieces directly support the claim? Is it one weak observation or multiple strong indicators? (4) Make judgment: If multiple relevant, strong pieces of evidence with no contradictions = claim well supported. Evidence quality checklist: STRONG evidence is (1) Specific ("bubbles formed" not "something happened"), (2) Quantitative when possible ("temperature rose 15°C" better than "got hot"), (3) Reproducible ("happened in all three trials" better than "happened once"), (4) Directly relevant (addresses the claim directly), (5) Not explainable by alternatives (gas from reaction, not from boiling).
A student claims: "A chemical reaction occurred when solid A was mixed with liquid B."
Evidence/observations recorded:
-
Bubbles formed immediately and continued for about 30 seconds.
-
The mass of the closed container stayed the same (within $\pm 0.01,\text{g}$).
-
The mixture changed from clear to cloudy.
-
The beaker felt cold to the touch.
Which statement best evaluates the claim using the evidence?
The claim is supported only because the beaker felt cold; bubbling and cloudiness are not relevant to chemical reactions.
The claim is not supported because the mass did not change, so no chemical reaction could have occurred.
The claim is not supported because the mixture became cloudy, which proves it was only physical mixing.
The claim is supported because gas formation (bubbling) and a new appearance (cloudiness) are indicators of a chemical reaction; the other observations do not contradict it.
Explanation
This question tests your ability to evaluate whether evidence adequately supports scientific claims about chemical reactions, distinguishing relevant from irrelevant evidence and assessing whether evidence is sufficient for the claim. Supporting scientific claims requires three things: (1) RELEVANCE—does the evidence actually relate to the claim? If claiming "reaction is exothermic," evidence about temperature increase is relevant, but evidence about color is not (color doesn't indicate exo vs endo). (2) SUFFICIENCY—is there enough evidence? Single weak observation usually insufficient; multiple strong observations or reproducible quantitative data provide sufficient support. (3) QUALITY—is the evidence specific and measurable? "Temperature increased by 12°C" is stronger than "it got warm." For claim "chemical reaction occurred," relevant sufficient evidence might include gas production + temperature increase + precipitate formation (three indicators of new substances), while just "substances mixed" would be insufficient (mixing doesn't prove reaction). The student's claim that a chemical reaction occurred is well-supported by multiple strong indicators: bubbling (gas formation) shows new gaseous products formed, cloudiness indicates precipitate formation or new suspended particles, and the cold feeling suggests an endothermic reaction absorbing heat—all three are classic signs of chemical change, while constant mass simply confirms conservation of mass in a closed system. Choice A correctly evaluates the evidence by identifying that gas formation and appearance change are strong indicators of chemical reaction, and noting that the other observations (constant mass and temperature change) don't contradict the claim—in fact, they provide additional support. Choice B incorrectly assumes mass must change in chemical reactions, when actually mass is conserved in closed systems; Choice C wrongly dismisses bubbling and cloudiness as irrelevant when they're actually key indicators; Choice D mistakenly claims cloudiness proves only physical mixing when it often indicates precipitate formation in reactions. The evidence evaluation framework: (1) Read the claim carefully: What exactly is being claimed? (2) For EACH piece of evidence, ask: Does this relate to the claim? (Relevance test), Does this indicate what the claim says? (Support test), Could this observation happen WITHOUT the claim being true? (Alternative explanation test). (3) Count relevant evidence: How many pieces directly support the claim? Is it one weak observation or multiple strong indicators? (4) Make judgment: If multiple relevant, strong pieces of evidence with no contradictions = claim well supported. If only weak or ambiguous evidence = claim not well supported. If evidence contradicts claim = claim not supported. Be honest about evidence quality!