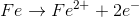Reaction Types
Help Questions
AP Chemistry › Reaction Types
When is the oxidation number of H (-1)?
Never
When it is bonded to a more electronegative element
When it is bonded to a less electronegative element
Only when it is bonded to Flourine
Only when it is bonded to Chlorine
Explanation
We typically think of Hydrogen as having an oxidation number of +1. However when it is bonded to a less electronegative element such as Na it is actually assigned an oxidation number of -1.
For the redox reaction shown, which of the following half reactions occurs in the anode?
Explanation
Recall that oxidation always occurs at the anode (in both the electrochemical and galvanic cells). 



If you have a solution that consists of a monoprotic acid (HA), with a pKa of 4.1 and at a pH of 5.8, what is the predominant species present?
HA
A-
H2A+
H3O+
Equal amounts of acid and conjugate base are present.
Explanation
Since pH > pKA, the deprotonated form of the acid is predominant.
Put the following acids in order of their INCREASING acid strength: HI, HCl, HBr, HF.
HI, HCl, HBr, HF
HF, HCl, HBr, HI
HI, HBr, HCl, HF
HF, HBR, HI, HCl
HCl, HBr, HI, HF
Explanation
Larger halogen size leads to greater acidity because of weaker H-X interactions.
Consider the following reaction:
What is the oxidizing agent, and what is the reducing agent?
Silver is the oxidizing agent and copper is the reducing agent
Silver is the reducing agent and copper is the oxidizing agent
Silver is the oxidizing agent and there is no reducing agent
No redox chemistry occurs
Silver is the reducing agent and there is no oxidizing agent
Explanation
Let's break down the reaction into two separate reactions:

We can see that copper loses electrons, while silver gains electrons. Recall that oxidation is loss and reduction is gain, with regard to electrons. Copper is oxidized and silver is reduced.
However, this question asks for the oxidizing agent and reducing agent. Recall that the oxidizing agent is reduced, while the reducing agent is oxidized. Since copper is oxidized, it is the reducing agent. Similarly, since silver is reduced, it is the oxidizing agent.
Which of the following is what determines the strength of an acid?
The Ka
The Kb
Its physical state
How many bonds the central atom makes
Electronegativity values
Explanation
The Ka is the acid dissociation constant, and thus it is what determines how strong the acid is. Stronger acids dissociate to a greater extent and produce lower pH values.
In the following reaction, which compound is being oxidized?
Copper
Sulfur
Hydrogen
Nitrate
No oxidation takes place
Explanation
Hydrogen doesn't change. Cu2+ doesn't change (partnered with S2- then with SO42-). Sulfur goes from S2- and S6+(paired with 6 O2- with a 2– charge), showing an oxidation. Nitrogen goes from N5+ to N2+ meaning it was reduced.
How many electrons are involved in the following reaction?
1 e-
2 e-
4 e-
5 e-
10 e-
Explanation
The common factor between 2 e- and 5 e- is 10. Therefore the number of electrons involved is 10 e-.
A buffer using acetic acid (pKa=4.76) is titrated with NaOH. What is the pH at half the equivalence point?
2.38
4.76
7.00
9.52
12.36
Explanation
The pH at half the equivalence point is equal to the pKa of the acid.
Put the following acids in order of their INCREASING acid strength: HI, HCl, HBr, HF.
HI, HCl, HBr, HF
HF, HCl, HBr, HI
HI, HBr, HCl, HF
HF, HBR, HI, HCl
HCl, HBr, HI, HF
Explanation
Larger halogen size leads to greater acidity because of weaker H-X interactions.