Enthalpy
Help Questions
AP Chemistry › Enthalpy
H2O ice melts to liquid H2O \[Heat is added\]
Which of the following is correct?
I. Δ H is positive
II. ΔS is positive
III. ΔH is negative
IV. ΔS is negative
I & II
I only
II only
I and III
I and IV
Explanation
When heat is added, enthalpy increases, ΔH refers to enthalpy
ΔS refers to entropy, which is disorder; disorder increases as it moves from solid to liquid to gas state.
As ice goes to liquid water, entropy increases
Calculate ΔH for the following reaction:
CH4 (g) + O2 (g) ⇌ CO2 (g) + H2O (l)
Compound ΔH
CH4 (g) -74.8 kJ/mol
H2O (l) -285.8 kJ/mol
CO2 (g) -393.5 kJ/mol
-890.3 kJ/mol
889.7 kJ/mol
not enough information
-604.5 kJ/mol
604.5 kJ/mol
Explanation
ΔH = Σ ΔHf products - Σ ΔHf reactants
ΔHf O2 or any element is 0
First step is to balance the equation:
CH4 (g) + O2 (g) ⇌ CO2 (g) + 2H2O (l)
ΔH = Σ ΔHf products - Σ ΔHf reactants
= \[-393.5 kJ/mol + 2(-285.8) kJ/mol\] - (-74.8 kJ/mol)
= -890.3 kJ/mol
Which of the following is a true statement regarding the energy involved in the formation of Methane, CH4, from graphite C(s) and H2(g) ?
Energy will be released
Energy will be consumed
Energy will be released, then consumed
Energy will be consumed, then released
None of the other answers
Explanation
This problem requires a knowledge of the energy involved when bonds are broken versus when bonds are formed. Energy is released when bonds are formed, and energy is consumed when bonds are broken. Here, methane is being formed from C and H2. Since the carbon source, graphite, is monoatomic the carbon will be involved only in the formation of C–H bonds on it's way to forming methane. The H however exists as H2 so bonds will need to be broken to allow for C–H bonds to form. So first energy is consumed to break the H–H bonds, it is then released when the C–H bonds form.
The formation of nitrogen dioxide is a two step process.


The net reaction is 
What is the change in enthalpy when creating four moles of nitrogen dioxide?
Explanation
Hess's law states that the change in enthalpy for a total reaction can be considered equal to the sum of the enthalpy changes for every step involved in the reaction. In other words, we can determine the enthalpy change for nitrogen dioxide by adding the enthalpy changes for both steps involved in its formation.
This gives us the total change in enthalpy for the listed reaction, 
Which of the following statements is true concerning a chemical reaction?
The 

A catalyst reduces the enthalpy change for the reaction
Exothermic reactions are always spontaneous
Endothermic reactions have lower activation energies than exothermic reactions
Explanation
When a chemical reaction is represented graphically, we see that the enthalpy change is reversed between the forward and reverse reactions. If a reaction produces energy in a forward process, it will require an input of energy in the reverse process, and vice versa.
A catalyst only affects the rate of a chemical reaction; it does not affect the equilibrium. Finally, exothermic reactions are not always spontaneous, but will have lower activation of energies compared to endothermic reactions.



What is the change in enthalpy for the given reaction?
Explanation
The change in enthalpy is calculated by:
When 



Since the reactions are in the correct order, adding all the 




What is the enthalpy of the following reaction?
Explanation
The change in enthalpy is calculated by:
When 



It is important to first balance the reaction before performing calculations. The coefficients are important in determining the change in enthalpy of a reaction. For this particular reaction, since there are two moles of product, the enthalpy of formation for 



What is the change in enthalpy for the following reaction?
Explanation
The change in enthalpy is calculated by:
When 



It is important to first balance the reaction before performing calculations. The coefficients are important in determining the change in enthalpy of a reaction.



What is the change in enthalpy for the following reaction?
Explanation
The change in enthalpy is calculated by:
When 



It is important to first balance the reaction before performing calculations. The coefficients are important in determining the change in enthalpy of a reaction.
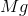

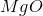
What is the change in enthalpy for the following reaction?
Explanation
The change in enthalpy is calculated by:
When 
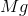

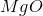
It is important to first balance the reaction before performing calculations. The coefficients are important in determining the change in enthalpy of a reaction.











































































