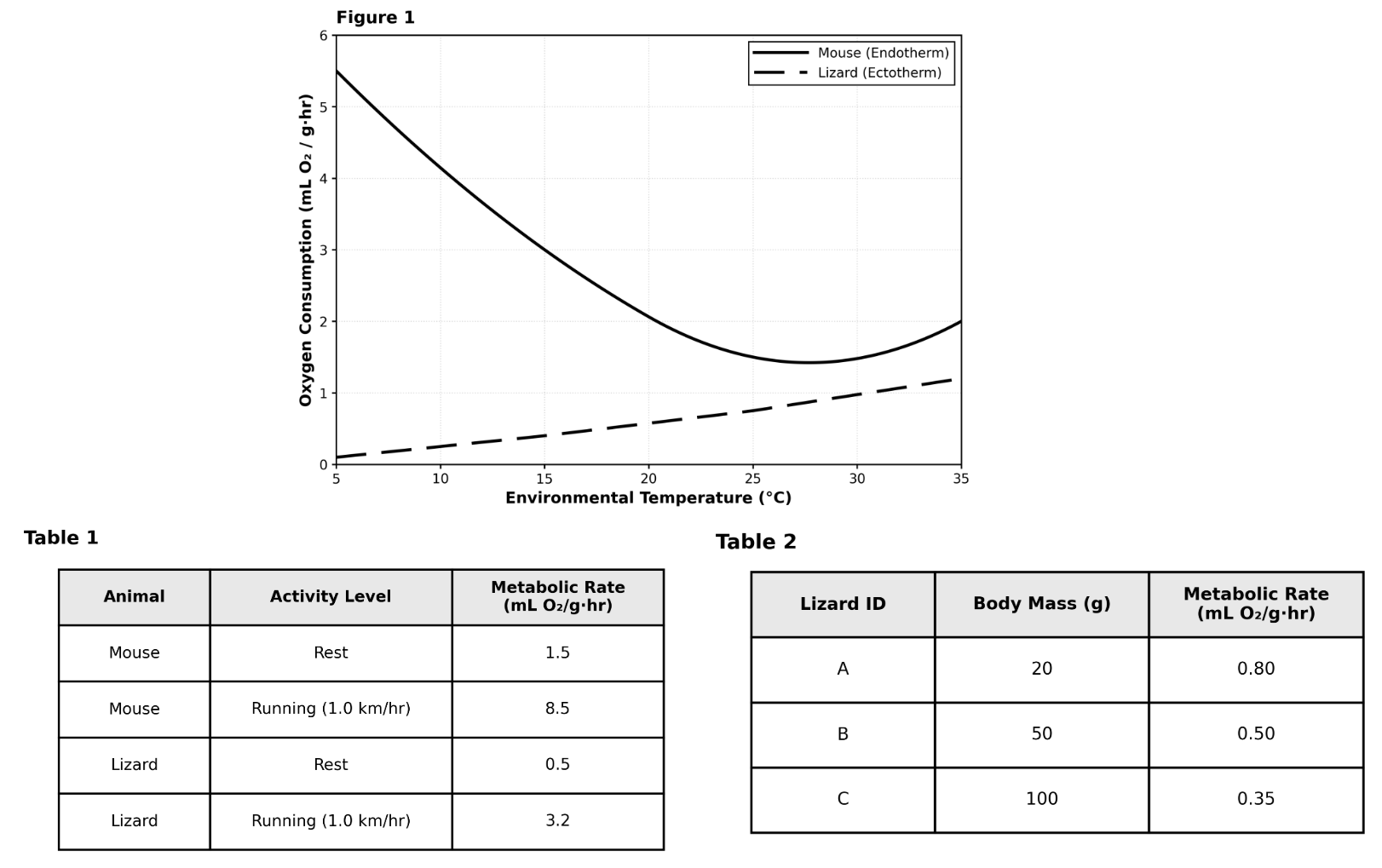
Evaluating Trends & Making Predictions Practice Test
•15 QuestionsPASSAGE IV
GEOPHYSICS: This passage is adapted from a study on the structure of Earth’s interior using seismic waves.
Seismologists study the interior of the Earth by analyzing the propagation of seismic waves generated by earthquakes. There are two main types of body waves:
•P-waves (Primary waves): Compressional waves that travel through solids, liquids, and gases.
•S-waves (Secondary waves): Shear waves that travel only through solids.
The velocity of these waves depends on the density and physical state (solid or liquid) of the material they travel through. Abrupt changes in velocity indicate boundaries between Earth's layers.
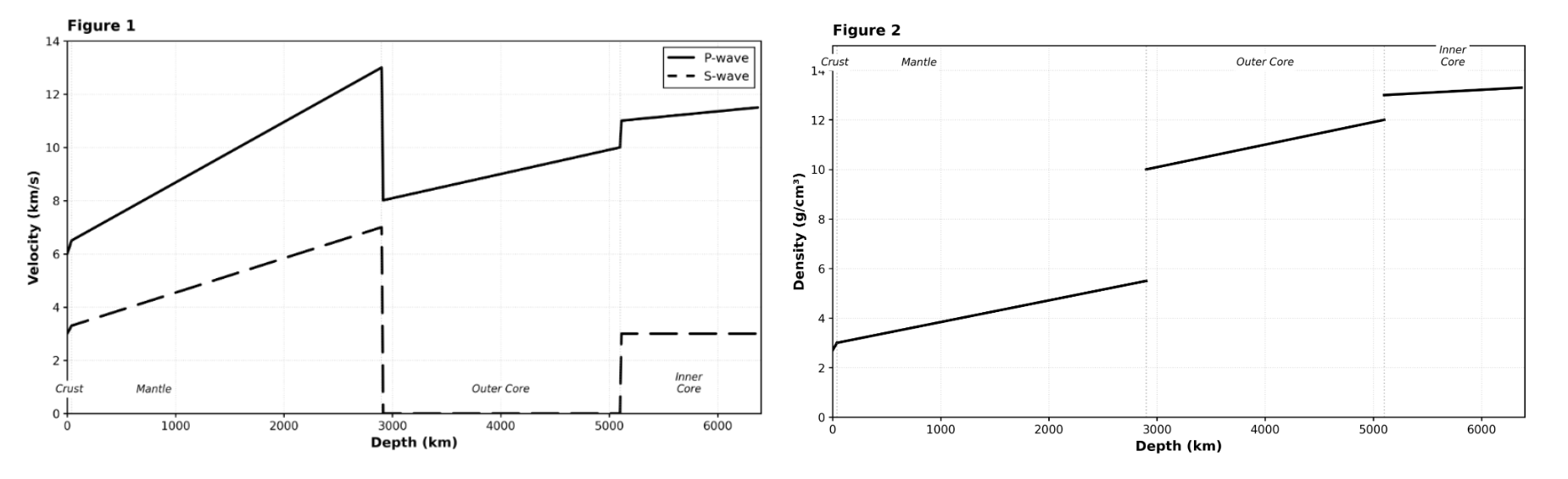
Which of the following statements best describes the relationship between depth and density within the Mantle (0-2,900 km), according to Figure 2?
Which of the following statements best describes the relationship between depth and density within the Mantle (0-2,900 km), according to Figure 2?