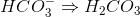Identifying and Defining Acids and Bases
Help Questions
SAT Subject Test in Chemistry › Identifying and Defining Acids and Bases
Questions 1 - 1
1
Which of the following cannot act as a Bronsted-Lowry base (proton recipient) in aqueous solution?
Explanation



AI TutorYour personal study buddy
Question of the DayDaily practice to build your skills
FlashcardsStudy and memorize key concepts
WorksheetsBuild custom practice quizzes
AI SolverStep-by-step problem solutions
Learn by ConceptMaster concepts step by step
Diagnostic TestsAssess your knowledge level
Practice TestsTest your skills with exam questions
GamesLearn through free games