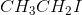Organic Chemistry
Help Questions
Organic Chemistry › Organic Chemistry
What is the IUPAC name for the following compound?

Explanation
The longest carbon chain is 







Identify the given organic functional group.

Aldehyde
Carboxylic acid
Acid anhydride
Ketone
Ester
Explanation
Aldehydes are carbonyls with one R-group and one hydrogen attached to the carbonyl carbon.
Ephedrine (shown below) contains what type of amine?

Secondary
Primary
Tertiary
Quaternary
Neutral
Explanation
A secondary amine is an amine (nitrogen atom) that is attached to two carbon-containing groups (alkyl groups or aryl groups). The nitrogen in ephedrine is attached to two alkyl groups, making it a secondary amine.
Primary amines are generally written as 

Identify the given organic functional group.

Aldehyde
Carboxylic acid
Acid anhydride
Ketone
Ester
Explanation
Aldehydes are carbonyls with one R-group and one hydrogen attached to the carbonyl carbon.
This molecule contains a carbonyl functional group. One substituent of the carbonyl carbon is a hydrogen. What is this molecule?
Aldehyde
Ketone
Acid
Amino acid
Explanation
A carbonyl functional group takes the form of 
An acid contains the carboxylic acid functional group, which takes the form of 

Amino acids contain the carboxylic acid functional group (


Assuming that this molecule is planar, determine whether this molecule is aromatic, and name the number of 

Not aromatic, 8 
Aromatic, 8 
Aromatic, 6 
Non aromatic, 6 
Explanation
There is no p orbital surrounding the Boron atom, so the ring does not have a fully conjugated pi system. In addition, there are 8 
What is the major product of the following reaction?
None of these
Explanation
The reaction shown is a Markovnikov addition of a hydracid (HX) across a double bond. According to Markovnikov's rule, the hydrogen gets added to the lesser-substituted carbon in the double bond, and the halide (in this case, 

Which of the following compounds can be considered a Lewis base?
Explanation
A Lewis base is a compound that donates a pair of electrons. The only compound shown that possesses an electron pair that can be donated is ammonia. 


Which of the following substrates would have the fastest reaction rate for an SN1 mechanism?
Explanation
The SN1 mechanism involves the formation of a carbocation intermediate in the rate-determining step. The most stable carbocation will produce the fastest reaction. We can immediately eliminate any answer choices that will produce primary or secondary carbocations, since a tertiary carbocation will be much more stable. When comparing tertiary carbocations, larger and more electronegative substituents will allow for more charge stabilization.
Since the tertiary carbocation formed by the dissociation of iodide from 
Which statement is not true regarding stereoisomers?
Corresponding stereoisomers have the same biological properties
Corresponding stereoisomers have the similar physical properties except for the rotation of light
Corresponding stereoisomers have the same molecular formulas
Diastereomers are easier to separate than enantiomers
Explanation
Corresponding stereoisomers differ in biological properties. The inversion of just one stereocenter on a large, complex molecule can alter the biological properties of the entire molecule. All other statements are true.