Principles of Oxidation-Reduction Reactions
Help Questions
AP Chemistry › Principles of Oxidation-Reduction Reactions
Consider the following reaction:
What is the oxidizing agent, and what is the reducing agent?
Silver is the oxidizing agent and copper is the reducing agent
Silver is the reducing agent and copper is the oxidizing agent
Silver is the oxidizing agent and there is no reducing agent
No redox chemistry occurs
Silver is the reducing agent and there is no oxidizing agent
Explanation
Let's break down the reaction into two separate reactions:

We can see that copper loses electrons, while silver gains electrons. Recall that oxidation is loss and reduction is gain, with regard to electrons. Copper is oxidized and silver is reduced.
However, this question asks for the oxidizing agent and reducing agent. Recall that the oxidizing agent is reduced, while the reducing agent is oxidized. Since copper is oxidized, it is the reducing agent. Similarly, since silver is reduced, it is the oxidizing agent.
In the following reaction, which compound is being oxidized?
Copper
Sulfur
Hydrogen
Nitrate
No oxidation takes place
Explanation
Hydrogen doesn't change. Cu2+ doesn't change (partnered with S2- then with SO42-). Sulfur goes from S2- and S6+(paired with 6 O2- with a 2– charge), showing an oxidation. Nitrogen goes from N5+ to N2+ meaning it was reduced.
Consider the following reaction:
Which of the following statements is true?
Zinc is oxidized and copper is reduced
Zinc is reduced and copper is oxidized
Sulfur is oxidized and copper is reduced
Sulfur is reduced and copper is reduced
Explanation
For any oxidation-reduction reaction, the oxidized atom will lose electrons and the reduced atom will gain electrons. Zinc is originally in elemental form, so it has an initial oxidation state of zero. As a product, it has a charge of 
Copper originally has an oxidation state of 
Sulfur remains in the sulfate complex ion, which retains its overall 
Which of the following reactions involves the transfer of electrons?
Redox
Neutralization
Decomposition
Combination
Explanation
Redox reactions occur when one species is oxidized—loses electrons—and another is reduced—gains electrons. Thus, this is the only one of the given options that involves the transfer of electrons.
Oomycetes of the genus Phytophthora are capable of causing economic damage by destroying crops. The name Phytopthora from the Greek phyton = plant, and phthora = destruction literally means plant-destroyer. However, chemists may battle their destructive effects.
The compound named copper(II) carbonate can be used to destroy the plant-destroyer, and can be produced as shown in the following equation:
What is the net ionic equation for the given reaction that produces copper(II) carbonate?
Explanation
An ionic equation is a chemical equation in which ionic compounds in an aqueous solution are written as dissociated ions.
A complete ionic equation shows all the ions present in the solution (which includes both the reactive and spectator ions), as the dissociated ions. The complete ionic equation for this problem's reaction is:
It is worth noting that in a complete ionic equation, it becomes readily apparent which ions are involved in the oxidation-reduction reaction, and which ions are the spectator ions (those that do not change during the reaction). For example since 




The net ionic equation shows only the reactive ions in solution and the product(s) formed by them. Therefore a net ionic equation can most simply be made by looking at the complete ionic equation and omitting the spectator ions as shown below:
Net ionic equation:
Which statement about the reaction equation is correct?



This is a combustion reaction
This is a hydrolysis reaction
Explanation




Which statement about this reaction is FALSE?
Chromium is oxidized during the reaction
Aluminum is oxidized during the reaction.
Chromium is the oxidizing reagent for this reaction.
This reaction is an example of a single displacement reaction.
Explanation
In Cr2O3, Cr has a charge of +3. The chromium metal product is neutral, and therefore has a charge of 0. So, Cr is reduced from +3 to 0.
Remember, OIL RIG. Oxidation Is Loss (of electrons) and Reduction Is Gain (of electrons).
In this reaction, electrons are transferred from to .
calcium . . . hydrogen
chlorine . . . hydrogen
hydrogen . . . calcium
calcium . . . chlorine
hydrogen . . . chlorine
Explanation
A transfer of electrons indicates an oxidation-reduction reaction. The element losing electrons is oxidized, while the element gaining electrons is reduced.
Calcium is initially a neutral solid, but as a product has a charge of 
Hydrogen initially has a charge of 
We can thus conclude that calcium was oxidized and hydrogen was reduced, indicating that electrons were transferred from calcium to hydrogen.
The charge on chlorine remains 
Methane combusts in the presence of oxygen according to the following reaction:
Which of the following statements is true concerning the reaction?
Carbon is oxidized in the reaction
Oxygen is oxidized in the reaction
Carbon has an initial oxidation state of
Oxygen has a charge of 

Explanation
By comparing the oxidation number of an atom as a reactant and its oxidation number as a product, we can determine if the atom has been oxidized or reduced. In increase in oxidation number indicates a loss of electrons, or oxidation. A decrease in oxidation number signals a gain of electrons, or reduction.
For electrochemistry, you should familiarize yourself with the traditional oxidation states of hydrogen 



Carbon is initially in the form of methane, meaning that it is attached to four hydrogen atoms. The molecule is neutral, and each hydrogen has an oxidation number of 

In the a product, carbon is attached to two oxygens, each with a charge of 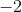

Since carbon went from an oxidation state of 
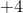
What does oxidation mean?
Loss of electrons
Gain of electrons
Loss of protons
Gain of protons
Loss of neutrons
Explanation
Think "OIL RIG" Oxidation Is Loss of electrons, Reduction Is Gain of electrons. The loss and gain of electrons is what changes the charge of the atom or molecule. In redox reactions, electrons are transferred. Changing the number of protons changes the identity of the element ,which is not the case. Since chemical reactions involve valence electrons, the nucleus is unchanged.















