Energy Diagrams
Help Questions
AP Chemistry › Energy Diagrams
A catalyst is added to a reaction whose uncatalyzed energy diagram is shown. Which change would be expected for the catalyzed pathway, relative to the uncatalyzed pathway?
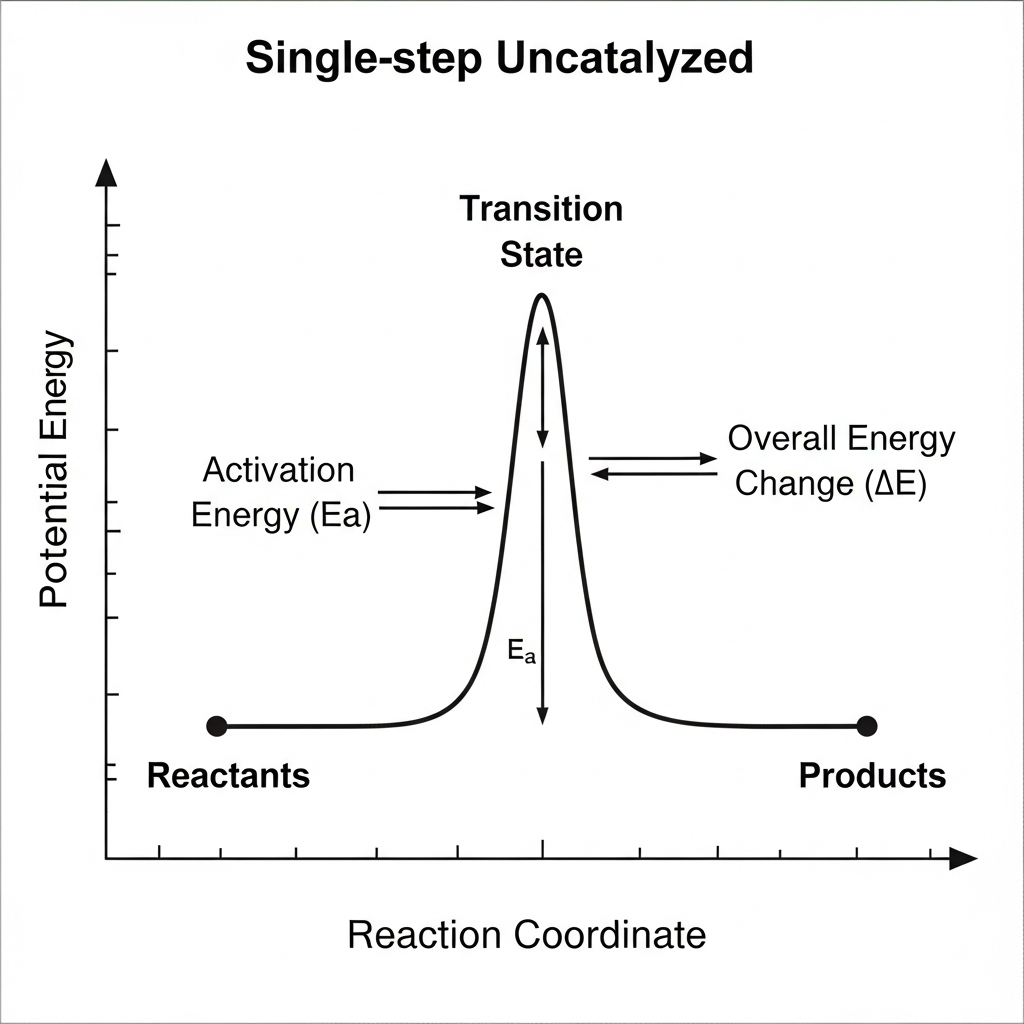
Product energy increases, making $\Delta H$ more positive
The horizontal distance between reactants and products decreases
Reactant energy decreases, making $\Delta H$ more negative
The peak increases because catalysts add energy to the reaction
The highest peak decreases while reactant and product energies stay the same
Explanation
This question tests understanding of how catalysts affect reaction energy diagrams. A catalyst provides an alternative reaction pathway with a lower activation energy, which appears on the diagram as a lower peak (or series of lower peaks) compared to the uncatalyzed reaction. Crucially, catalysts do not change the energy levels of reactants or products, so the starting and ending points remain the same, preserving the same ΔH for the reaction. The catalyst only affects the pathway between reactants and products, lowering the highest energy barrier that must be overcome. A common misconception (choice E) is thinking catalysts add energy to increase the peak, but catalysts actually lower activation energy by providing a more favorable pathway. When analyzing catalyzed reactions, verify that reactant and product energies remain unchanged while the transition state peak(s) decrease.
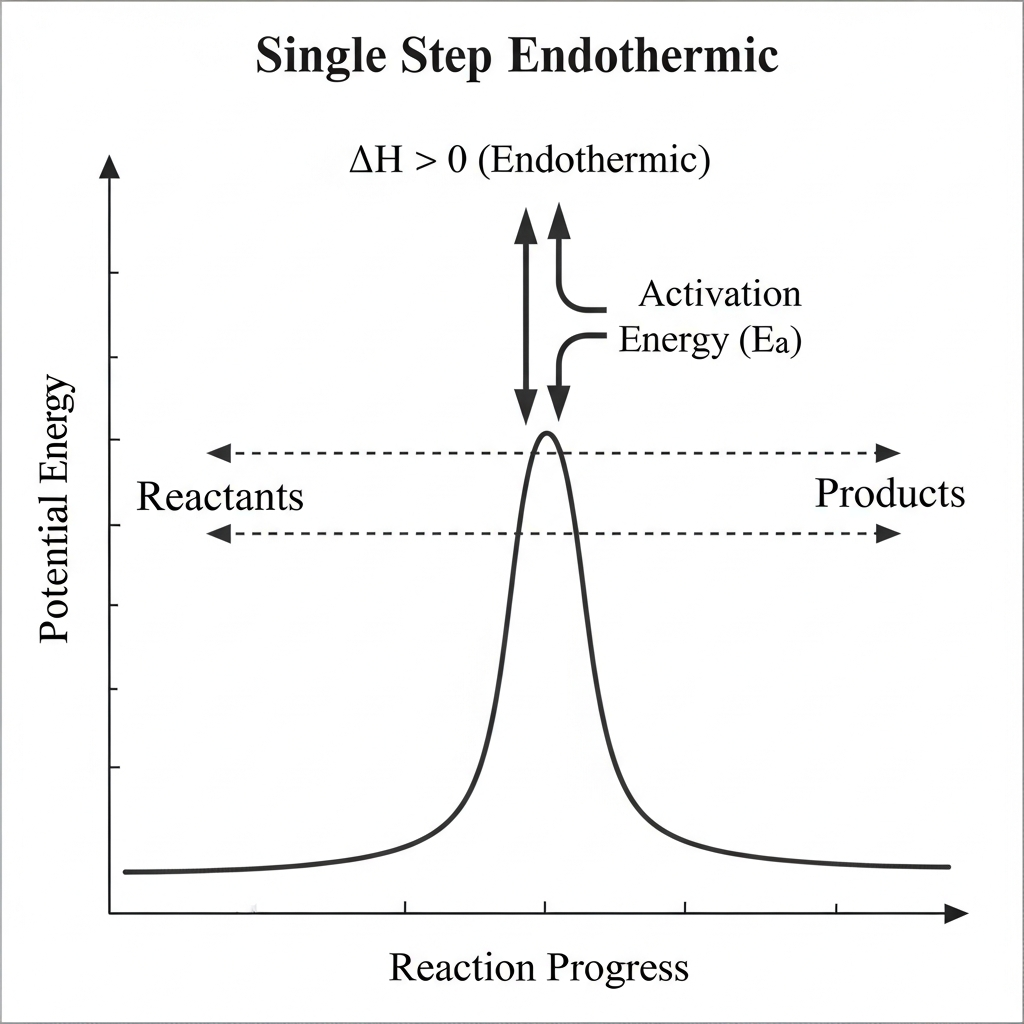
For the single-step reaction shown in the energy diagram, which statement best describes the sign of $\Delta H$?
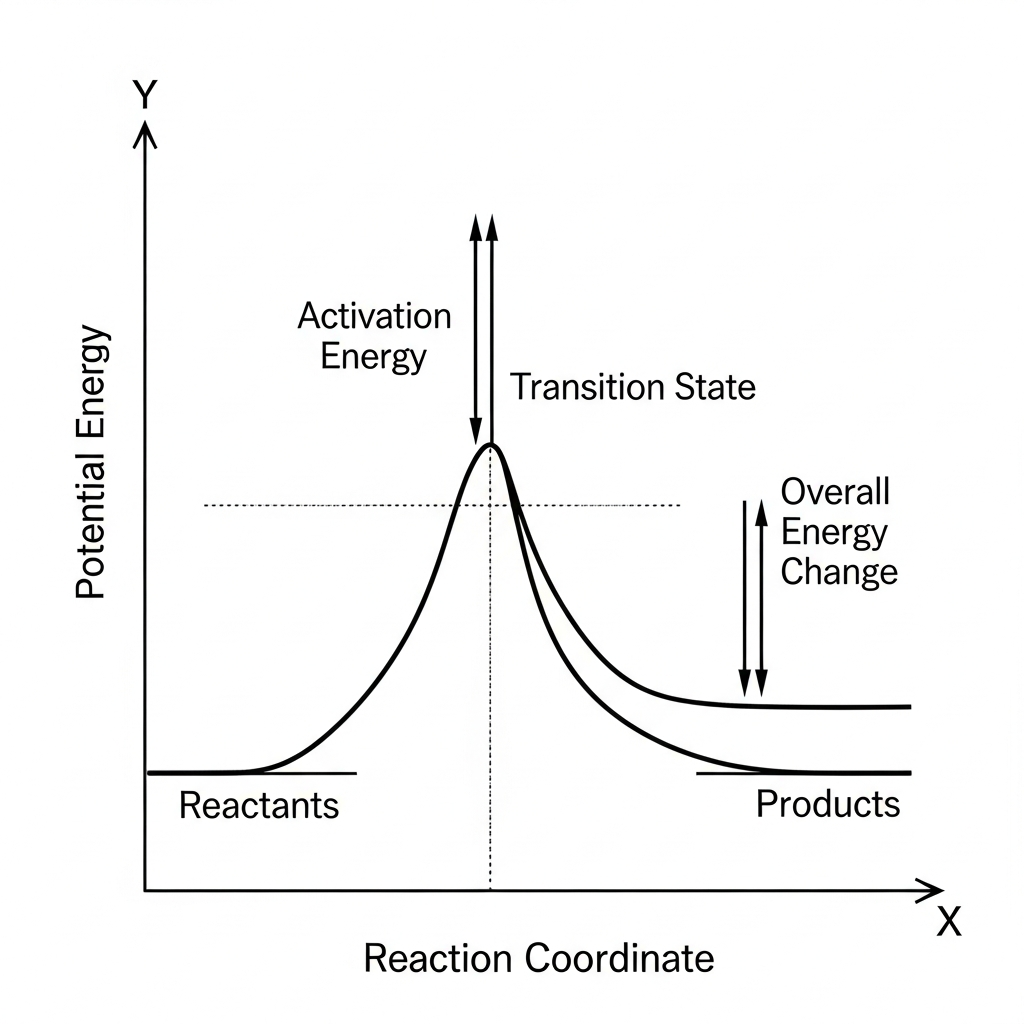
$\Delta H=0$ because there is only one transition state
$\Delta H<0$ because products are at higher potential energy than reactants
$\Delta H>0$ because products are at higher potential energy than reactants
$\Delta H<0$ because the peak is above the reactants
$\Delta H>0$ because the activation energy is positive
Explanation
This question tests the ability to determine the sign of ΔH from an energy diagram. The enthalpy change (ΔH) is determined by comparing the potential energy levels of reactants and products: if products are higher in energy than reactants, energy must be absorbed, making the reaction endothermic (ΔH > 0). The diagram shows products at a higher potential energy level than reactants, confirming that ΔH > 0. The presence of an activation energy peak is irrelevant to the sign of ΔH; all reactions have activation energy regardless of being endothermic or exothermic. A common misconception (choice E) is misreading the diagram and thinking ΔH < 0 when products are higher, which would violate energy conservation. When determining ΔH from an energy diagram, focus only on the vertical difference between reactant and product energy levels, ignoring transition states.
The energy diagram shown represents a reaction. Compared with the forward reaction, the reverse reaction has which relationship for activation energy and enthalpy change?
Reverse has smaller $E_a$ and $\Delta H>0$
Reverse has larger $E_a$ and $\Delta H<0$
Reverse has the same $E_a$ and $\Delta H=0$
Reverse has larger $E_a$ and $\Delta H>0$
Reverse has smaller $E_a$ and $\Delta H<0$
Explanation
This question tests understanding of the relationship between forward and reverse reactions on an energy diagram. Since the diagram shows products at a higher energy level than reactants, the forward reaction is endothermic (ΔH > 0), and the reverse reaction is exothermic (ΔH < 0 for reverse). The activation energy for the reverse reaction is the vertical distance from products up to the transition state peak, while for the forward reaction it's from reactants to the same peak. Because products are higher than reactants, the reverse reaction has a larger vertical distance to climb to reach the transition state, giving it a larger activation energy. A common misconception (choice A) is thinking the reverse reaction of an endothermic process would have a smaller activation energy, but this ignores the actual energy levels shown. When analyzing reverse reactions, remember that ΔH changes sign and activation energies depend on the vertical distances from starting points to the transition state.
Two pathways for the same reaction are shown: Pathway 1 has two steps (two peaks), and Pathway 2 has one step (one peak). Both have the same reactants and products. Which statement is correct?
(Vertical axis: potential energy; horizontal axis: reaction coordinate.)
Both pathways have $\Delta H=0$ because they share endpoints.
Pathway 2 must be faster because it is single-step.
Pathway 1 must be slower because it has more steps.
Pathway 1 has a different $\Delta H$ because it has an intermediate.
Whichever pathway has the lower highest peak has the lower overall $E_a$.
Explanation
This question tests the skill of comparing activation energies and rates for alternative reaction pathways in energy diagrams. The overall Ea for each pathway is determined by the height of its highest peak relative to reactants, with the pathway having the lower highest peak expected to be faster, regardless of the number of steps. This is because the rate is limited by the largest barrier, and both pathways share the same ΔH since they have identical reactants and products. Thus, option C correctly states that whichever pathway has the lower highest peak has the lower overall Ea. A tempting distractor is option A, which claims Pathway 1 is slower because it has more steps, but this is incorrect due to the misconception that the number of steps directly determines rate, ignoring activation energy heights. To compare pathways, evaluate the maximum Ea for each, as the lowest maximum barrier indicates the kinetically favored route.
A reaction proceeds in two steps with an intermediate I, as shown.
Energy
| R______/____
| / \ ____
| / ____ ____ P
| I
+--------------------------------> Reaction coordinate
Which step has the larger activation energy?
Both steps, because the two peaks are at the same height above the baseline.
Step 1 (R to I), because the first peak is higher above R than the second peak is above I.
Step 2 (I to P), because products are lower than reactants.
Step 2 (I to P), because the second peak is closer to P on the x-axis.
Neither step, because activation energy is the energy difference between R and P.
Explanation
This question tests the skill of comparing activation energies in multi-step reactions using relative peak heights. Step 1 has the larger Ea because its peak is higher above R than the second peak is above I, where Ea is step-specific from starting energy to transition state. The diagram shows two peaks with the first taller from R, and overall P below R for exothermicity. This identifies the rate-determining step as the one with the highest barrier. A tempting distractor is choice D, which incorrectly says both steps have equal Ea due to similar peak heights, ignoring relative starting points. Calculate each step's Ea from its own baseline to peak for accurate comparisons.
A two-step energy diagram is shown. Which step is the rate-determining step (slow step) based on the diagram?
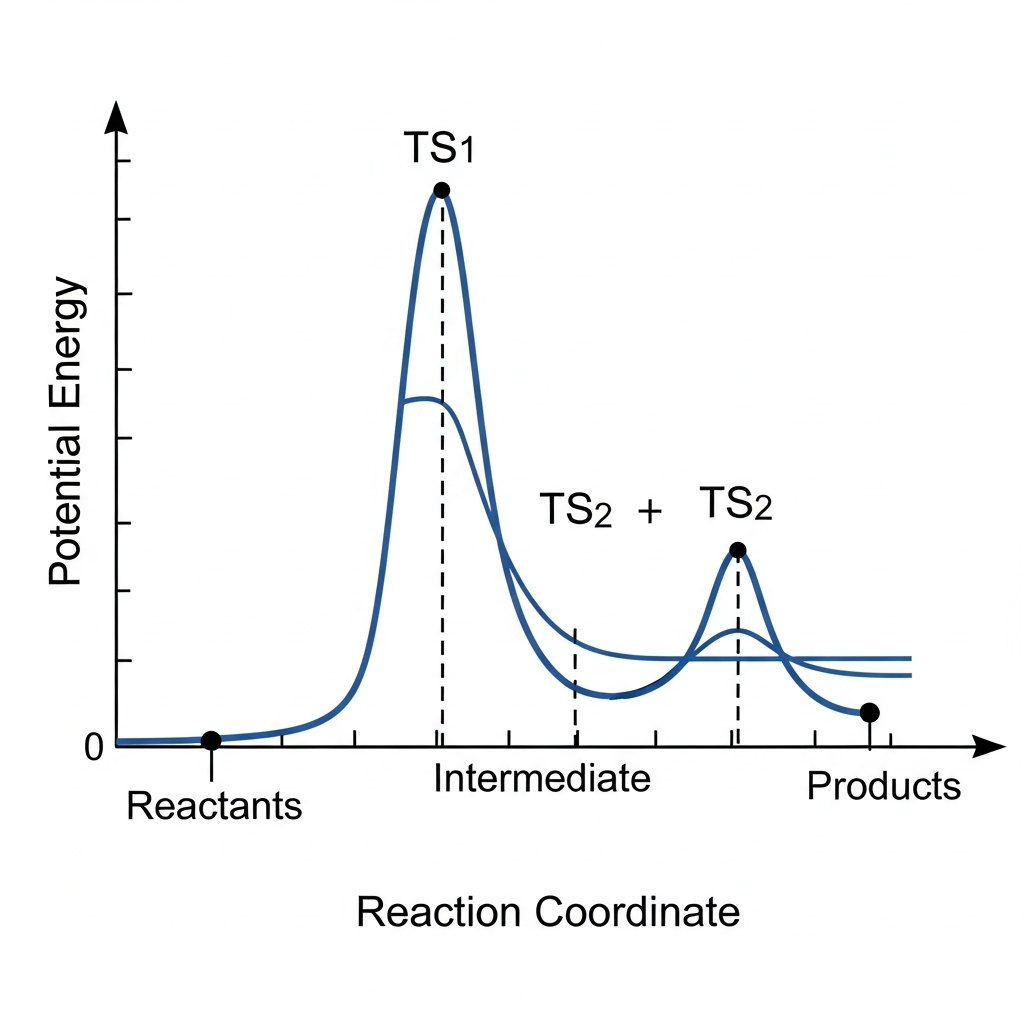
Step 2, because products are lower in energy than reactants
Neither; both steps are equally slow because there are two peaks
Step 1, because $E_a$ from reactants to TS1 is larger than from intermediate to TS2
Step 2, because the intermediate is lower than the reactants
Step 1, because $\Delta H$ for the overall reaction is negative
Explanation
This question tests understanding of rate-determining steps in multi-step reactions. The rate-determining step is the slowest step, which has the largest activation energy barrier. From the diagram, Step 1 has a larger activation energy (vertical distance from reactants to TS1) than Step 2 (vertical distance from intermediate to TS2), making Step 1 the rate-determining step. The overall ΔH of the reaction or the relative positions of intermediates and products do not determine which step is slowest; only the activation energy barriers matter. A common misconception (choice B) is thinking that the step leading to lower-energy products must be slower, but reaction rate depends on activation energy, not thermodynamics. To identify the rate-determining step, measure the activation energy for each step and select the one with the largest barrier.
Two pathways for the same reaction are shown in the energy diagram: Pathway 1 (uncatalyzed) and Pathway 2 (catalyzed). Which statement is consistent with the diagram?
Pathway 2 has a larger $E_a$ because it has more steps
Both pathways have the same $E_a$ because they start at the same reactant energy
Pathway 2 has the same $\Delta H$ as Pathway 1 but a smaller overall $E_a$
Pathway 1 has a smaller $E_a$ because its peak is lower than Pathway 2
Pathway 2 has a different $\Delta H$ because catalysts change product energy
Explanation
This question tests understanding of how catalysts affect reaction energy diagrams. A catalyst provides an alternative pathway with a lower activation energy but does not change the thermodynamics of the reaction, so ΔH remains the same for both pathways since they have the same starting and ending energy levels. Pathway 2 (catalyzed) shows multiple smaller peaks instead of one large peak, with the highest peak of Pathway 2 being lower than the single peak of Pathway 1, indicating a smaller overall activation energy. The overall activation energy is determined by the highest energy barrier that must be overcome, not the sum of all barriers. A common misconception (choice C) is that catalysts change the energy of products, but catalysts only affect kinetics, not thermodynamics. When comparing catalyzed and uncatalyzed pathways, verify that reactants and products are at the same energy levels in both diagrams, then identify the highest peak in each pathway to compare activation energies.
Two reactions, A and B, are shown on separate energy diagrams. Reaction A is exothermic with a high peak; Reaction B is endothermic with a lower peak. Which reaction is expected to be faster at the same temperature, based only on the diagrams?
(Vertical axis: potential energy; horizontal axis: reaction coordinate.)
Reaction A, because its products are lower in energy.
Reaction A, because exothermic reactions are always faster.
They must have the same rate because both are single-step.
Reaction B, because its activation energy barrier is lower.
Reaction B, because $\Delta H>0$ increases the rate.
Explanation
This question tests the skill of comparing reaction rates based on activation energies from energy diagrams. Reaction B has a lower peak, indicating a smaller activation energy barrier compared to Reaction A's high peak, even though Reaction A is exothermic and B is endothermic. According to transition state theory, the reaction rate is primarily determined by the height of the activation energy barrier, with lower Ea leading to faster rates at the same temperature. Therefore, option B correctly states that Reaction B is expected to be faster due to its lower activation energy. A tempting distractor is option A, which claims Reaction A is faster because exothermic reactions are always faster, but this is incorrect due to the misconception that thermodynamics (ΔH) directly dictate kinetics, whereas rate depends on Ea. When comparing reactions via energy diagrams, prioritize the activation energy height over the overall ΔH to predict relative rates.
Two energy diagrams represent two different reactions.
Reaction X:
Energy
| /\
| R___/ _____ P
+----------------->
Reaction Y:
Energy
| /\
| R_________/ _____ P
+----------------->
Which statement is supported by the diagrams?
Reaction Y has a smaller $E_a$ than Reaction X because its peak is higher.
Reaction X has a larger $E_a$ than Reaction Y because its peak is taller.
Reaction X has a larger $E_a$ than Reaction Y because its products are lower.
Both reactions have the same $E_a$ because both are single-step.
Reaction X has a smaller $E_a$ than Reaction Y because its peak is closer to R.
Explanation
This question tests the skill of comparing activation energies between different reactions using energy diagrams. Reaction X has a taller peak above its reactants compared to Reaction Y's shorter peak, indicating X has a larger Ea, as Ea is the vertical energy difference from R to the transition state. Both diagrams show P below R, suggesting exothermic reactions, but the key comparison is the barrier height. This demonstrates that reactions with similar thermodynamics can have different kinetics based on Ea. A tempting distractor is choice B, which wrongly claims X has smaller Ea due to peak proximity to R, based on the misconception that horizontal position affects energy barriers. When comparing diagrams, measure vertical heights from R to peaks for Ea, ignoring x-axis positioning.
Two different reactions, X and Y, have energy diagrams shown (each single-step). Reaction X has products much lower than reactants; reaction Y has products slightly lower than reactants. Which comparison is supported by the diagrams?
Reaction Y has a more negative $\Delta H$ than reaction X.
Reaction X has a more negative $\Delta H$ than reaction Y.
Reaction X has a larger forward $E_a$ than reaction Y because $\Delta H$ is more negative.
Both reactions have the same $\Delta H$ because both are exothermic.
Reaction X must have a smaller reverse $E_a$ than reaction Y because products are lower.
Explanation
This question tests the ability to compare thermodynamic properties between different reactions using energy diagrams. The enthalpy change (ΔH) is determined by the vertical distance between reactants and products - a larger drop means a more negative (more exothermic) ΔH. Since reaction X shows products much lower than reactants compared to reaction Y's slight drop, reaction X has a more negative ΔH. The activation energies and ΔH values are independent properties - a more negative ΔH does not automatically mean a larger Ea(forward). A common misconception is thinking all exothermic reactions have the same ΔH (choice E), but the magnitude of energy release varies with the specific vertical drop on the diagram. When comparing reactions, examine the relative positions of reactants and products separately from the transition state heights.